What’s All the Fuss About EMG?
By Bret Contreras and Andrew Vigotsky
In recent months, the subject of electromyography (EMG) has become somewhat controversial in the online fitness community. In fact, some fitness writers have even gone so far as to call EMG worthless and inherently flawed, but we completely disagree. The backlash against EMG is predictable if you understand the nature of the fitness industry. When new topics, methods, exercises, or systems gain rapid popularity, they tend to get scrutinized to a much greater degree. Several online authors including Bret Contreras and Brad Schoenfeld have been heavily promoting their EMG research and experiments over the past few years, and this has caused various fitness enthusiasts to question the validity of EMG. Questioning is always a good thing, so we encourage discussion pertaining to EMG. However, it is important to be scientifically grounded or else we won’t reach a consensus.
First off, we feel that the term worthless should be used sparingly in sports science. When one decides to cross the line and become an actual scientist, he is held to a higher standard. One can rarely ever say that something well researched is worthless, since almost everything has a purpose and provides some value depending on the situation. This applies not only to most exercises and most tools and instruments, but also to things that tend to be shunned upon in various fitness circles such as unstable surface training (UST), static stretching, and whole body vibration (WBV) training. For example, UST is valuable in various rehab settings, static stretching is the method of choice for increasing flexibility through neural and not architectural means, and WBV is promising for the promotion of tendon healing. Ignoring the research pertaining to these phenomenon and making bold statements about how overrated they are is commonplace in the S&C blogosphere, but you won’t see good scientists going down this path, nor will you see solid sports scientists say that EMG is worthless. Top sports scientists including Stu McGill, Paul Swinton, Stu Phillips, Brad Schoenfeld, and Jacob Wilson utilize EMG in their research for good reason – it allows them quantify the level of myoelectrical activity elicited during exercise.
To sports scientists, some of the recent claims surrounding EMG are puzzling to say the least, but others are understandable and reasonable. Like any tool, EMG has definite limitations, but that does not invalidate all EMG data. Rather, the problems lie within the interpretation, extrapolation, and generalization of the data rather than the data itself. It should be mentioned that all sports science tools have inherent limitations, including force plates (their accuracy is improved when adding a linear position transducer (LPT), and adding two LPT’s is even better), isokinetic dynamometers (maximum torque doesn’t always match up with what’s seen in dynamic exercises), ultrasound (it’s reliant on the force and angle of application by the examiner), and musculoskeletal modeling (it relies upon numerous assumptions). A good researcher learns how to work around these limitations and take necessary precautions in order to help him answer the questions he pursues. We happen to highly value EMG, and we’d like to share our knowledge and experiences with the readers.
The ideas presented in this article represent our interpretation of the current data utilizing our working knowledge of electromyography (EMG). There are incredibly intelligent scientists, electrical engineers, and biomedical engineers (e.g., Carlo De Luca, Dario Farina, Roger Enoka, George Dimitrov, and Nonna Dimitrova to name a few) who have dedicated their entire careers to studying EMG. These folks are the true experts in EMG. Quite frankly, there is currently a considerable amount of knowledge pertaining to EMG that the authors of this article (Bret and Andrew) do not yet understand. We have only been using and studying EMG for a few years, not decades like our colleagues, nor have we made it our primary passion or focus of research. However, similar to a car, a television, or a microwave, one does not have to possess a world-class understanding of all of the inner-workings and nuances in order to use them. There are entire journals that are dedicated to EMG and neuromuscular physiology (e.g., Journal of Electromyography & Kinesiology, Muscle & Nerve, etc.), and the science can be quite intimidating and overwhelming. Don’t believe us? Here are a few examples of how complex it gets: (Detecting the Unique Representation of Motor-Unit Action Potentials in the Surface Electromyogram, Conduction Velocity of Quiescent Muscle Fibers Decreases During Sustained Contraction, Adjustments Differ Among Low-Threshold Motor Units During Intermittent, Isometric Contractions). Try making full sense of these papers! There is a lot more to EMG than what we know, but this also applies to those who frequently critique it – they’re in the dark about much of this stuff too, just like we are!
Learn by Doing
Like most things, one gains tremendous knowledge about EMG through experimentation and not just reading research. We have found EMG to be straightforward and intuitive, but from time to time we’ve encountered issues. For example, sometimes when collecting data you just get wacky readings and you have to figure out what’s going on – you change out the electrode, pull the lead out and reconnect it, restart the unit, etc. Most of the time this will solve the issue. Moreover, technology goes bad over time, so wires need to be replaced, electrodes can’t be allowed to expire, etc. When you gain sufficient experience, you know what looks right and what doesn’t – you watch the individual perform a movement and you examine the waveform on the computer to make sure it looks legit. Every once in a while, you don’t catch the glitch right away and you end up having to exclude the data from your study, but this is rare. In warmer climates such as ours (Phoenix, AZ), sweating can be a serious issue in terms of causing electrodes to slide around. In this case, special adhesive spray can be purchased to improve the stickiness without interfering with the electrodes. We’ve also learned little tricks to improve upon our methods, for example taping or strapping down the wires while providing slack so the wires don’t pull on the electrodes. We suspect that many of those who criticize EMG would learn to appreciate and utilize EMG in their decision making if they had access to EMG and gained sufficient experience with it.

EMG, Force Plate, Motion Capture, and Modeling at Stu McGill’s Lab
What Research Still Hasn’t Sufficiently Investigated…
Research hasn’t yet mapped out EMG-joint angle curves for each muscle and joint action. Some muscles activate fairly consistently throughout their range of motion, others activate highest at their shortest ROMs and least when stretched, and others vice-versa. What does this mean for hypertrophy training? We don’t know yet. Range of motion is a good thing based on hypertrophy research, so this implies that just going for the highest activation isn’t the end-all-be-all for hypertrophy, as one could just perform isometrics at the joint angle that maximizes EMG activation. When considering muscular hypertrophy, one needs to consider the 3 primary mechanisms: mechanical tension, metabolic stress, and muscle damage. Tension (muscle force) is well correlated with muscle activation during isometric contractions when not under fatigue, but dynamic (especially explosive) contractions and fatigue muddies the waters. One can impose high levels of metabolic stress without attaining high peak activation, as it’s more related to constant tension above a certain threshold of muscle activation. Moreover, one can impose high levels of muscle damage without attaining high mean or peak activation, as it’s more related to strain/stretch than muscle activation. Based on this knowledge, a variety of movements should always be performed for maximizing the hypertrophic response. That said, you won’t do well in your pursuits to grow a muscle if you’re not regularly highly activating it!
What is EMG?
Electromyography measures the electrical activity of muscles during exercise. While EMG doesn’t directly measure muscular tension, the two should be very similar (although slightly off-set), as the electrical activity that EMG measures is simply a measurement of the nervous system’s signal to the muscles, combined with some detection of electric activity from peripheral factors in the muscles. However, it is important to note that muscles can produce active and passive elastic force – active force will require myoelectric activity but passive elastic force just requires a stretch. Increased EMG activity is indicative of the nervous system’s attempt to produce more muscular force, or to compensate for force losses related to fatigue. With gross movements and surface muscles, surface EMG is fairly reliable, but with fine-motor movements and deep muscles, wire EMG is required for accurate estimates.
The Typical Arguments
As for the arguments that have been cropping up over the Internet as of late, well, some of them have some scientific basis (just grossly overstated), while others are just plain silly. Here are the common arguments listed below.
Cadavers Detect EMG, Proving that it’s Useless – Allegedly Nautilus inventor Arthur Jones hooked a cadaver up to EMG and detected activity.
Don’t get us started with Arthur Jones. He was an amazing inventor but a horrendous scientist. Any sport scientist worth his salt would cringe when listening to Jones ramble. Case in point, here is what Jones had to say about EMG:
“And, please, do not suggest that we can answer that question by using EMG readings; you can wipe your ass on EMG readings, all they produce is a bunch of lines on a piece of paper that nobody can understand. About twenty-four years ago, we wired up a dead man and then produced EMG results by moving his limbs; readings that damned sure did not result from muscular function. The only worthwhile result I have ever seen from EMG tests was one which clearly established the existence of internal muscular friction: the amplitude of the reading was greater during positive work, and much lower during negative work with the same level of resistance; because, while lifting the weight the muscles had to overcome both the resistance and their own internal friction, while the friction helped them when the weight was being lowered back down.
But, of course, no scientist would ever agree with that above statement since none of them are even aware that muscles have friction, and would deny it if you suggested it to them. After all, it was not their discovery, thus it obviously cannot be true. And then you wonder why I consider scientists, as a group, to be the dumbest people on the planet? Exceptions? Almost certainly; and someday I hope to meet one.”

Arthur Jones: Great Inventor, Angry & Crummy Scientist
This clearly elucidates Jones’ arrogance and ignorance. Hundreds of research scientists can easily understand EMG, so he’s incorrect about that. We personally don’t believe that a cadaver elicits noticeable EMG activity and that Jones simply didn’t control for movement artifacts, but let’s give him the benefit of the doubt and say it does. How many microvolts does moving a cadaver’s limbs register? We suppose that the EMG would be detecting static electricity. Would it elicit 5 microvolts? 20 microvolts? During MVIC testing, depending on the muscle, it’s not uncommon to detect over 1,000 microvolts of EMG activity. If 5-20 microvolts were indeed detected with the cadaver, then static electricity still wouldn’t greatly interfere with myoelectrical readings since it would only amount to 0.5-2.0% of the total. Since Jones never published his findings or explained his methods, we’re left guessing. We have tested living subjects while passively moving their limbs around, and no EMG activity was detected, therefore, we’re calling bullshit on Jones.
Titin: A Winding Filament
To further elaborate on what an imbecile Jones was, we’d like to point out that he was incorrect about his assertion pertaining to muscular friction. The decreased EMG seen during eccentric contractions compared to concentric contractions is attributed mainly to the mechanics of titin – a large molecule that contributes significant amounts of passive elastic force to activated, lengthened muscular contractions. This phenomenon is very well researched thanks to some excellent researchers examining titin mechanics, including Walter Herzog, Henk Granzier, Tim Leonard, Kiisa Nishikawa, Stan Lindstedt, and Paul LaStayo. Click on these links to learn more about this topic:
When active muscles lengthen: properties and consequences of eccentric contractions
Is titin a ‘winding filament’? A new twist on muscle contraction
Eccentric Exercise: Many questions unanswered
Titin force is enhanced in actively stretched skeletal muscle
Mechanisms of enhanced force production in lengthening (eccentric) muscle contractions
As you can see, scientists aren’t “the dumbest people on the planet” – Jones was guilty of confirmation bias and the Dunning-Kruger Effect. He also realized, just as many of today’s experts have, that being incredibly bold and condescending while ridiculing everyone and calling them stupid garners much attention and develops a cult like following of blind, raving fans.
Want some more gems from Jones? Check this video out:
Articles citing Jones’ opinions don’t hold much weight to legitimate sports scientists, but we digress…
Surface EMG is Invalid – surface EMG (sEMG), at best, only reads the superficial muscle fibers or only tells you if the muscle is “on”.
This argument implies that there is a drastic difference in activation between the deep and superficial fibers and that changes in amplitude are not indicative of alterations in activation. However, numerous studies have shown that, as expected, sEMG has a positive correlation with fine wire EMG for larger skeletal muscles (we would expect large muscles like the gluteus maximus to be the most highly correlated) such as gluteus medius (Semciw, Neate, & Pizzari, 2014), quads and hamstrings (Jacobson, Gabel, & Brand, 1995), lower limb muscles (Chapman, Vicenzino, Blanch, Knox, & Hodges, 2010), and positive correlations with force output, velocity, and fatigue. It is true that fibers along the surface tend to consist of a greater percentage of fast-twitch fibers while deeper fibers tend to consist of a greater percentage of slow-twitch fibers (Dickx et al., 2010), but we don’t feel that this drastically alters the EMG signal, especially during traditional strength training experiments. Some smaller, deep muscles such as those of the rotator cuff aren’t well correlated with fine wire EMG (Allen, Brookham, Cudlip, & Dickerson, 2013; Rajaratnam, Goh, & Kumar, 2014; Waite, Brookham, & Dickerson, 2010)., but others such as certain abdominal wall muscles, the psoas, and quadratus lumborum are well correlated (McGill, Juker, & Kropf, 1996).
Moreover, EMG is actually an outstanding method for quantifying muscle activation. While simple palpation can tell you if a muscle is working or not, EMG provides actual data. A researcher can report the data in microvolts, or alternatively, a researcher can normalize the data to a maximum voluntary isometric contraction (MVIC) in order to gauge how hard the muscle is working relative to maximum capacity. Finally, one can also normalize the data to a reference movement such as a bodyweight squat. A researcher can examine mean (average) or peak (maximum) activation, or focus on the area under the curve by looking at integrated EMG (iEMG). Each of these tells the researcher unique things, and one can also simply examine the waveform to attain a pretty accurate picture as to what’s going on during the exercise, especially if motion or video capture is simultaneously recorded and synced with the EMG data.
There are ways to improve the validity of sEMG, such as using multiple electrodes (recent technology has hundreds of mini-electrodes lined up to allow the researcher to examine the entire length of muscles and detect more activity). This technology improves sEMG technology by roughly 10% (Farina, Merletti, & Enoka, 2014).
Technologies similar in nature to sEMG are regularly used in the clinical setting, including EKG (electrocardiograph) and EEG (electroencephalograph). Doctors depend on these to monitor electrical activity in order to make diagnoses, evaluate patient progress, and prevent catastrophe. If these arguments against sEMG are true, then how can we depend on these medical technologies?
Fine Wire EMG is Painful and Invalid – fine wire EMG is painful, unethical, and it would be extraordinarily difficult to find volunteers to participate in a fine wire EMG study. If you were able to get subjects, it would be too painful for the subject to give a meaningful/proper contraction. Furthermore, it’s highly probable that the subject will damage his/her muscle and/or nerves by doing so.
It’s fairly obvious that anyone who would cite this argument has no experience with fine wire EMG. In the video below, we place a fine wire electrode in the distal portion of Bret’s bicep brachii, and he seems to be doing just fine. Does it feel a bit uncomfortable? Sure. But so does wearing knee wraps or a belt during heavy squats. This doesn’t invalidate the data, especially if the subjects are warmed up properly. Burgar et al. (1997) showed that placing fine wire electrodes in individuals’ index finger muscles did not affect force output. It should also be noted that investigators had a 93% success rate in placing the electrodes in 60 different muscles – we think it’s safe to assume that this number would be even greater in muscles of interest to sports scientists (hamstrings, quadriceps, glutes, pecs, lats, biceps, triceps, deltoids, etc.). Lastly, there have been numerous studies that utilize fine wire electrodes that not only received IRB approval (ethics), but also were able to recruit plenty of subjects. Here are 10 random studies that we pulled up; researchers don’t seem to have a problem finding subjects or attaining ethics approval.
| Study utilizing fine wire EMG | n |
| Nuber, G. W., Jobe, F. W., Perry, J., Moynes, D. R., & Antonelli, D. (1986). Fine wire electromyography analysis of muscles of the shoulder during swimming. The American journal of sports medicine, 14(1), 7-11. | 11 |
| Németh, G., Kronberg, M., & Broström, L. Å. (1990). Electromyogram (EMG) recordings from the subscapularis muscle: description of a technique. Journal of orthopaedic research, 8(1), 151-153. | 12 |
| Ryan, M. M., & Gregor, R. J. (1992). EMG profiles of lower extremity muscles during cycling at constant workload and cadence. Journal of Electromyography and Kinesiology, 2(2), 69-80. | 18 |
| Sapsford, R. R., Hodges, P. W., Richardson, C. A., Cooper, D. H., Markwell, S. J., & Jull, G. A. (2001). Co‐activation of the abdominal and pelvic floor muscles during voluntary exercises. Neurourology and Urodynamics, 20(1), 31-42. | 7 |
| Malanga, G. A., Jenp, Y. N., Growney, E. S., & An, K. N. (1996). EMG analysis of shoulder positioning in testing and strengthening the supraspinatus.Medicine and science in sports and exercise, 28(6), 661-664. | 17 |
| Eastwood, P. R., Allison, G. T., Shepherd, K. L., Szollosi, I., & Hillman, D. R. (2003). Heterogeneous activity of the human genioglossus muscle assessed by multiple bipolar fine-wire electrodes. Journal of Applied Physiology, 94(5), 1849-1858. | 8 |
| Burgar, C. G., Valero-Cuevas, F. J., & Hentz, V. R. (1997). FINE-WIRE ELECTROMYOGRAPHIC RECORDING DURING FORCE GENERATION: Application to Index Finger Kinesiologic Studies1. American journal of physical medicine & rehabilitation, 76(6), 494-501. | 5 |
| Jacobson, W. C., Gabel, R. H., & Brand, R. A. (1995). Surface vs. fine-wire electrode ensemble-averaged signals during gait. Journal of Electromyography and Kinesiology, 5(1), 37-44. | 12 |
| Bigland-Ritchie, B., Johansson, R., Lippold, O. C., & Woods, J. J. (1983). Contractile speed and EMG changes during fatigue of sustained maximal voluntary contractions. J neurophysiol, 50(1), 313-324. | 16 |
| Andersson, E. A., Nilsson, J., & Thorstensson, A. (1997). Intramuscular EMG from the hip flexor muscles during human locomotion. Acta Physiologica Scandinavica, 161(3), 361-370. | 11 |
| TOTAL | 117 |
Here’s a video demonstrating fine wire electrode placement. You can feel it, but we don’t think it’s painful to the point where it markedly changes muscle activation. Wearing a tight belt, putting on tight knee wraps, or putting a heavy bar on the back can be painful during squatting, but obviously lifters can overcome this and highly activate their muscles.
MVIC is Not Truly Maximal – maximum voluntary isometric contraction, or MVIC, is not necessarily a true maximal contraction, you’d need to use the superimposed burst technique to know if you’re really maximally activating the muscle
MVIC is obtained by having the subject contract his/her muscle as hard as he/she can in an isometric fashion at the joint angle that is known to maximize muscle activation of the muscle being examined. This number is then used as a denominator in order to calculate EMG %MVIC:
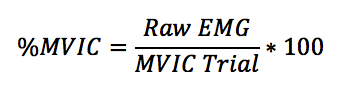
As one can see, no matter what the MVIC trial is, it’s not biased as all trials are divided by the same number from the MVIC trial. If a researcher tests the bench press, incline press, and decline press and normalizes the data to MVIC, each exercise gets divided by the same number. In this manner, it’s fair and unbiased.
The raw number gathered from the isometric MVIC trial is usually pretty high, but is not always higher than what that person can achieve during movement, especially heavy or explosive resistance training. Sure, in order to achieve true maximal contraction, one would need to use the superimposed burst technique, wherein investigators would stimulate the muscle to contract as hard as it physiologically can (it’s worth noting that sometimes the superimposed burst technique does not lead to increased activation over what an MVIC elicits). However, MVIC is named appropriately: maximal voluntary isometric contraction, and all it does is put raw EMG data (usually in mV or μV) into perspective. Normalizing to MVIC makes the data easier to digest and interpret, and it gives investigators and readers a frame of reference so different muscles can be compared to one another (i.e., intensity).
We could nitpick all day long. For example, we could tell someone that his 1RM isn’t really a 1RM as he could have probably done more if he was maximally psyched up and motivated and fed and rested. But we’re okay with going by the most weight he’s ever lifted, and we’re okay with comparing exercises by normalizing them to MVIC’s.
EMG is Unreliable
There are several types of reliability, but we are assuming this argument is referring to the test-retest reliability. EMG is no more than an expensive, highly sensitive voltmeter that measures the voltage (in mV or μV) under or around the electrode. Test-retest reliability refers to the variation in measurements taken by the same person under the same conditions. EMG reliability is equivocal and it’s dependent on which muscles are being measured and under what circumstances. For example, sEMG is not reliable for measuring serratus anterior activation levels during dynamic flexion, dynamic abduction, isometric flexion, isometric abduction, and in the bench press plus. However, sEMG on the serratus anterior is reliable for other movements (Hackett et al., 2014). The conditions under which EMG is used must be tested in order to ensure reliability, as reliability is not something that can be generalized.
EMG is Invalid
This depends on the claim. Does EMG accurately depict myoelectric activity? Of course it does. Does it accurately predict metabolic stress or muscle damage? Not always. Does it accurately depict muscle force? It depends on contraction velocity and action, and also fatigue (Hof, 1997; Korner et al., 1984; Lawrence & De Luca, 1983; Milner-Brown & Stein, 1975; Onishi et al., 2000; Perry & Bekey, 1981). Does it correlate perfectly with hypertrophy? No, but it is definitely related to hypertrophy.
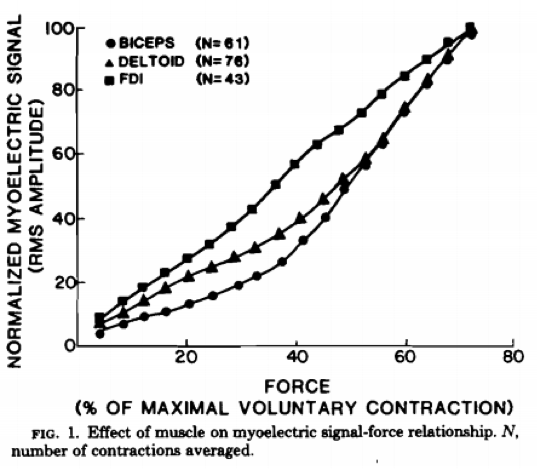
From: Myoelectric signal versus force relationship in different human muscles by Lawrence & DeLuca
Check out the chart above. As you can see, the relationship with EMG and muscle tension is fairly linear. We’ll take EMG over some guru’s opinion any day of the week.
MVIC is Unreliable – Different MVIC numbers can be collected on different trials
Though this may be the case, again, all data is divided by the same number. Frequently, many researchers perform multiple MVIC trials (3-5) and average them, which helps to correct for the possible variability in MVIC trials. Moreover, a study is more accurate when all the exercises are performed in one trial and all the data is divided by the same MVIC measurement. If data is obtained on multiple testing occasions, then problems could arise due to the electrode placement, MVIC measurement, etc. However, most studies obtain their EMG data on the same testing occasion, so this usually isn’t a problem.
EMG is Improperly Used for Exercise Selection
Research reports averages. Researchers with vast experience with EMG notice that there is great individual variation with regards to motor control and muscle activation during resistance training. People utilize muscles in different ways, and everyone is unique. Therefore, applying “averages” to every person will be problematic, as we cannot assume that what applies to the average person applies to every individual. Some individuals show very high levels of muscle activation in exercise A but not exercise B, whereas other individuals will show very high levels of muscle activation in exercise B but not exercise A. This is where the principle of individualization applies, and ideally every individual would be able to experience comprehensive EMG testing to see exactly how their muscles activate, identify which exercises lead to the highest levels of mean and peak activation in each of the major muscles, identify which exercises didn’t pan out very well, and identify the loading protocols that maximize muscle activation. However, this is rarely if ever the case, so we have to combine published EMG knowledge with anecdotal feedback, palpation, and other means.
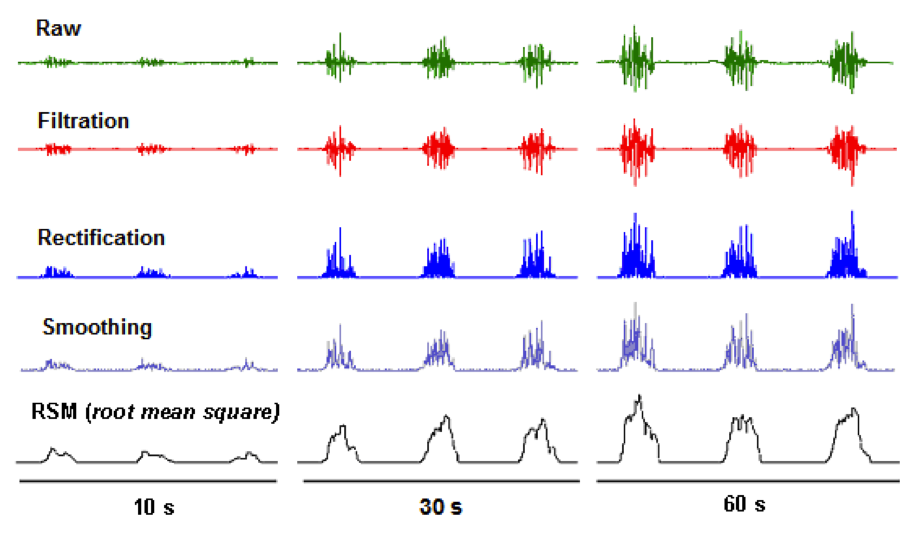
EMG Yields Messy Data – When you look at EMG data, there are spikes and noise
These spikes/noise can be alleviated using complex algorithms (filtering, smoothing, and RMS), which help to make the data more digestible.
EMG Doesn’t Tell you about Posture or Form
It’s not supposed to. For this, one should utilize motion capture in order to assess the position of different joints. A thermometer doesn’t tell you blood pressure, but this doesn’t mean the thermometer is useless. Just as a doctor utilizes a variety of tools for information about a patient’s health, a fitness professional should rely upon a variety of tools for information about an exercise or movement’s efficacy (more on this later).
Muscle Activation is not Directly Correlated with Muscle Protein Synthesis (MPS) or Hypertrophy
This is a good point. However, although not directly correlated with MPS and hypertrophy, EMG is correlated with fMRI as being an accurate measure of activation (Adams et al., 1992; Dickx et al., 2010), and we know from research that fMRI activation is a good predictor of hypertrophy and MPS (Wakahara et al., 2012; Wakahara et al., 2013). Therefore, we don’t think it’s a stretch to say that EMG could very well be a good predictor of MPS and hypertrophy.
Limitations
As scientists that utilize EMG, we are definitely aware of its limitations. Four years ago, Bret wrote a blog about EMG, Here are most of them listed below:
- Some deep muscles are impossible to measure with surface EMG and require fine-wire EMG
- Some individuals suck at maximally contracting muscles isometrically, which will yield especially higher percentages during experiments when standardized according to MVC
- Some muscles are harder to maximally contract voluntarily than others, which will also yield higher percentages during experiments when standardized according to MVC
- MVC is inconsistent and varies from trial to trial, as even when you try to use the exact electrode placement and contract as hard as possible during MVC, it almost always yields different levels of MVC (this explains why the most reliable approach is to study all of the exercises in one session)
- MVC is joint-angle dependent; one has to know the best MVC positioning for each muscle (which can vary from individual to individual)
- Electrode site placement is critical; improper placement can get interference from nearby muscles or body parts, or worse, it could measure the incorrect muscle
- Electrode angle placement should be parallel with muscle fibers
- External pressure from clothing can interfere with readings
- Electrodes can lose their stickiness and start sloughing off or fall off completely, which can yield improper readings (electrodes should be taped on ideally)
- Changes in muscle belly geometry can interfere with readings
- Sliding of the skin in which the electrodes are placed can move the recorded area and interfere with readings
- Explosive movements like sprints, plyometrics, jump squats, or Olympic lifts can sometimes yield unusually high peak EMG readings
- Wires can get in the way while performing exercises, sometimes getting caught and ripping electrodes off in the process, which will pretty much end the session as when you place the electrode back on your readings will be skewed (wires should be taped to the body or velcro straps should be used)
- Bumping an electrode during an exercise with the object used for resistance will throw the reading off considerably
- During one-rep maxes there is no averaging of reps, which diminishes reliability
- Choosing the interval period for the repetition is arbitrary
- Range of motion and type of contraction are not considered, thereby giving partial reps and isometric contractions an advantage
- Some individuals subconsciously flex and activate muscles during exercises which gives a false reading (you can squeeze the biceps as hard as possible on a squat; that doesn’t mean that they helped produce the motion)
- Form varies considerably from individual to individual, and form can be purposely altered to favor a certain muscle or muscle group
- Tissue characteristics can play a role in EMG readings, as some muscles are thicker than others and some muscles are located underneath fatty deposits
- Skin preparation can affect readings (and skin should be shaved & gel should be used)
- On a smaller scale, physiological and temperature conditions can alter readings
- Neighboring cross-talk between muscles can interfere with readings, as can external noise like power hums and faulty grounding
Practical Applications
Below lists the practical applications of EMG, including a video that elaborates on its utility for personal training. HERE is a Wikipedia link to EMG. EMG is used in the research in a number of ways:
- To compare muscle activation across different exercises
- To determine the regional activation (functional subdivisions) that exist within muscles
- To see the effects of tweaking exercise form on muscle activation
- To determine whether various methods such as unstable surface training or whole body vibration increase or decrease muscle activation
- To determine the isometric positions that elicit the highest activation, which are used for normalization purposes (called maximum voluntary isometric contractions, or MVIC’s)
- To examine the effects of cueing and attentional focus on muscle activation
- To examine muscle activation during bloodflow restriction training, high rep training to failure versus heavy weights, and special techniques such as pre-exhaustion, drop sets, and rest pause
- To examine the EMG-angle curve, peak activation time-points, and minimum activation time-points during exercises or sports activities
- To examine rate of EMG rise during heavy or explosive movement
- To examine muscle activation during sticking regions in the powerlifts
- To examine muscle activation differences in elite versus novice athletes
- For biofeedback training in rehabilitation
- To examine muscle onset times
- To examine whether myoelectric silence occurs (such as during stoop lifting)
- To examine differences in muscle activation in normal subjects versus subjects in pain
- To examine asymmetries
- To examine muscle activation differences between ideal form and poor form (such as knee valgus)
- To examine gender differences in muscle activation during various tasks
- To examine muscle activity during different phases or portions of a sporting task (such as the braking, propulsive, and swing phases in sprinting, along with the acceleration and maximum speed phases)
- To compare different types of activities (such as vertical jumps, horizontal jumps, and lateral jumps)
- To learn more about the bilateral deficit
- To examine the effects of increasing intensity of load or effort on muscle activation
- To examine the effects of fatigue on muscle activation
- To determine the level of cocontraction
- To help estimate muscle force, intraabdominal pressure, and spinal loading in modeling studies
- To determine increases in neural drive following training interventions
- To examine changes in agonist and antagonist muscle activation following various stretching and manual therapy protocols
- To control for muscle activation when measuring muscle stiffness
- To examine muscle activation in the elderly or in special populations, and to identify neuromuscular diseases or dysfunction such as muscular dystrophy, ALS, myasthenia gravis, or carpal tunnel syndrome
Below is a video detailing how Bret Contreras uses EMG in his personal training. He has found EMG to be highly useful in helping him achieve better results with his clients.
A Variety of Tools Should Always be Utilized
It should be mentioned that sports scientists utilize many tools and methods to help them answer questions, each of which possess inherent strengths and weaknesses. As previously mentioned, EMG is no exception; it definitely has its pros and cons. But the same can be said for every biomechanical tool, including force plates, accelerometers, linear position transducers, isokinetic dynamometers, force treadmills, MRI, and ultrasound. With regards to determining exercise efficiency, a good practitioner will rely upon a variety of information, some of which may include:
- Discussing exercise with fellow lifters, trainers, and coaches, attending seminars, and/or watching videos pertaining to resistance training
- Reading magazines, books, blog posts, and published literature pertaining to resistance training
- Performing an exercise and “feeling the burn”
- Intentional inducement of delayed onset muscle soreness (DOMS) by performing many sets of a certain exercise
- Muscle palpation during exercise performance
- Biomechanical analysis of the exercise in terms of movement patterns, joint angles, actions, and ROM’s, muscle lengths, muscle actions, muscle fiber origins, insertions, and lines of pull
- Biomechanical analysis of the exercise in terms of joint torques, moment arms, muscle forces, joint forces, and spinal loading (these require inverse dynamics and computer modeling)
- Surface and fine wire electromyography (EMG) activation (examining mean & peak along with the entire pattern)
- Magnetic Resonance Imaging (MRI) activation (including T2 weighted imaging immediately after the session to estimate activation or 48 hours afterward to estimate damage)
- Blood samples (examining levels of hormones, lactate, creatine kinase)
- Training one’s self and/or clients and examining the effects (even better if variables are controlled and the scientific method is utilized)
- Conducting longitudinal training studies and examining the training effects (this is where a number of tools can be used to examine gains in flexibility, strength, power, hypertrophy, speed, and/or stamina, which can include technology such as goniometers, ultrasound, tensiomyography (TMG), mechanomyography (MMG), computed tomography (CT scans), near-infrared spectroscopy (NIRS), motion capture, dual energy x-ray absorption (DXA), biopsies, isokinetic dynamometers, and force plates)
As you can imagine, each of these have inherent strengths, weaknesses, and limitations. One strength of EMG is that it gives you numerical data so you’re not relying solely on subjective feedback.
Wrapping it Up
Though EMG has its limitations, it is still a highly valuable tool that should be used, but not solely relied upon. It is not the end-all, since it doesn’t tell you vital information that is needed to accurately gauge an exercise’s effectiveness. However, it’s also not as useless as a frying pan for measuring muscle activation. Claiming so demonstrates serious ignorance.
Rather than looking to fitness experts with zero experience with research or sports science equipment for information about research methodology in strength training, we recommend looking to actual researchers. Individuals such as Stu Phillips, Brad Schoenfeld, Stu McGill, Jacob Wilson, David Behm, and Greg Lehman are all brilliant and respected researchers who have contributed to the fields of sports science and biomechanics. They understand EMG’s limitations, but continue to use it because they understand its potential value and applications. We will continue to utilize EMG for our experiments, and we believe that the main drawback surrounding EMG does not lie within EMG itself, but with the interpretation of its data.
References
Adams, G. R., Duvoisin, M. R., & Dudley, G. A. (1992). Magnetic resonance imaging and electromyography as indexes of muscle function.Journal of applied physiology (Bethesda, Md. : 1985), 73(4), 1578–83.
Allen, T. R., Brookham, R. L., Cudlip, A. C., & Dickerson, C. R. (2013). Comparing surface and indwelling electromyographic signals of the supraspinatus and infraspinatus muscles during submaximal axial humeral rotation. J Electromyogr Kinesiol, 23(6), 1343-1349. doi: 10.1016/j.jelekin.2013.08.002
Burgar, C. G., Valero-Cuevas, F. J., & Hentz, V. R. (1997). FINE-WIRE ELECTROMYOGRAPHIC RECORDING DURING FORCE GENERATION: Application to Index Finger Kinesiologic Studies. American journal of physical medicine & rehabilitation, 76(6), 494-501.
Chapman, A. R., Vicenzino, B., Blanch, P., Knox, J. J., & Hodges, P. W. (2010). Intramuscular fine-wire electromyography during cycling: repeatability, normalisation and a comparison to surface electromyography. J Electromyogr Kinesiol, 20(1), 108-117. doi: 10.1016/j.jelekin.2008.11.013
Dickx, N., Cagnie, B., Achten, E., Vandemaele, P., Parlevliet, T., & Danneels, L. (2010). Differentiation between deep and superficial fibers of the lumbar multifidus by magnetic resonance imaging. Eur Spine J, 19(1), 122-128. doi: 10.1007/s00586-009-1171-x
Dickx, N., D’Hooge, R., Cagnie, B., Deschepper, E., Verstraete, K., & Danneels, L. (2010). Magnetic resonance imaging and electromyography to measure lumbar back muscle activity. Spine, 35(17), E836–42.
Farina, D., Holobar, A., Gazzoni, M., Zazula, D., Merletti, R., & Enoka, R. M. (2009). Adjustments differ among low-threshold motor units during intermittent, isometric contractions. Journal of neurophysiology, 101(1), 350-359.
Farina, D., Merletti, R., & Enoka, R. M. (2014). The Extraction of Neural Strategies from the Surface Emg: An Update. J Appl Physiol (1985). doi: 10.1152/japplphysiol.00162.2014
Farina, D., Negro, F., Gazzoni, M., & Enoka, R. M. (2008). Detecting the unique representation of motor-unit action potentials in the surface electromyogram. Journal of neurophysiology, 100(3), 1223-1233.
Gazzoni, M., Camelia, F., & Farina, D. (2005). Conduction velocity of quiescent muscle fibers decreases during sustained contraction. Journal of neurophysiology, 94(1), 387-394.
Hackett, L., Reed, D., Halaki, M., & Ginn, K. A. (2014). Assessing the validity of surface electromyography for recording muscle activation patterns from serratus anterior. Journal of Electromyography and Kinesiology, 24(2), 221-227.
Herzog, W. (2014). Mechanisms of enhanced force production in lengthening (eccentric) muscle contractions. Journal of Applied Physiology, 116(11), 1407-1417.
Hof, A. L. (1997). The relationship between electromyogram and muscle force. Sportverletz Sportschaden, 11(3), 79-86. doi: 10.1055/s-2007-993372
Hoppeler, H., & Herzog, W. (2014). Eccentric Exercise: Many Questions Unanswered. Journal of Applied Physiology, 116(11), 1405-1406.
Jacobson, W. C., Gabel, R. H., & Brand, R. A. (1995). Surface vs. fine-wire electrode ensemble-averaged signals during gait. J Electromyogr Kinesiol, 5(1), 37-44.
Korner, L., Parker, P., Almstrom, C., Andersson, G. B., Herberts, P., Kadefors, R., . . . Zetterberg, C. (1984). Relation of intramuscular pressure to the force output and myoelectric signal of skeletal muscle. J Orthop Res, 2(3), 289-296. doi: 10.1002/jor.1100020311
Lawrence, J. H., & De Luca, C. J. (1983). Myoelectric signal versus force relationship in different human muscles. J Appl Physiol Respir Environ Exerc Physiol, 54(6), 1653-1659.
Lindstedt, S. L., LaStayo, P. C., & Reich, T. E. (2001). When active muscles lengthen: properties and consequences of eccentric contractions. Physiology,16(6), 256-261.
McGill, S., Juker, D., & Kropf, P. (1996). Appropriately placed surface EMG electrodes reflect deep muscle activity (psoas, quadratus lumborum, abdominal wall) in the lumbar spine. J Biomech, 29(11), 1503-1507.
Milner-Brown, H. S., & Stein, R. B. (1975). The relation between the surface electromyogram and muscular force. J Physiol, 246(3), 549-569.
Nishikawa, K. C., Monroy, J. A., Uyeno, T. E., Yeo, S. H., Pai, D. K., & Lindstedt, S. L. (2012). Is titin a ‘winding filament’? A new twist on muscle contraction. Proceedings of the Royal Society B: Biological Sciences,279(1730), 981-990.
Onishi, H., Yagi, R., Akasaka, K., Momose, K., Ihashi, K., & Handa, Y. (2000). Relationship between EMG signals and force in human vastus lateralis muscle using multiple bipolar wire electrodes. J Electromyogr Kinesiol, 10(1), 59-67.
Perry, J., & Bekey, G. A. (1981). EMG-force relationships in skeletal muscle. Crit Rev Biomed Eng, 7(1), 1-22.
Powers, K., Schappacher-Tilp, G., Jinha, A., Leonard, T., Nishikawa, K., & Herzog, W. (2014). Titin force is enhanced in actively stretched skeletal muscle.The Journal of experimental biology, 217(20), 3629-3636.
Rajaratnam, BS, Goh, JCH, & Kumar, VP. (2014). A Comparison of EMG Signals from Surface and Fine-Wire Electrodes During Shoulder Abduction. Int J Phys Med Rehabil, 2(206), 2.
Semciw, A. I., Neate, R., & Pizzari, T. (2014). A comparison of surface and fine wire EMG recordings of gluteus medius during selected maximum isometric voluntary contractions of the hip. J Electromyogr Kinesiol. doi: 10.1016/j.jelekin.2014.08.015
Waite, D. L., Brookham, R. L., & Dickerson, C. R. (2010). On the suitability of using surface electrode placements to estimate muscle activity of the rotator cuff as recorded by intramuscular electrodes. J Electromyogr Kinesiol, 20(5), 903-911. doi: 10.1016/j.jelekin.2009.10.003
Wakahara, T., Fukutani, A., Kawakami, Y., & Yanai, T. (2013). Nonuniform muscle hypertrophy: its relation to muscle activation in training session. Medicine and science in sports and exercise, 45(11), 2158–65.
Wakahara, T., Miyamoto, N., Sugisaki, N., Murata, K., Kanehisa, H., Kawakami, Y., … Yanai, T. (2012). Association between regional differences in muscle activation in one session of resistance exercise and in muscle hypertrophy after resistance training. European journal of applied physiology, 112(4), 1569–76.





It’s ironic i find this on my fb wall today. I just handed in my undergrad dissertation which included teaching myself how to use EMG. Lets just say mistakes were made in the process lol, but it gave me something else to talk about in the report. I also feel for you on the workload for you doctorate, this was a pretty brutal assignment, yours must have been mammoth in scale.
I decided to test the abdominal training, extension vs flexion. Interesting results, and definitley more work and research needed in a couple of areas I feel, but it’ll have to wait till i can get some post grad work done.
Hope the first submission goes well for you Brett. When do we get a sneak peek?
Epic write-up Bret, well done!
This is awesome Bret and Andrew, thank you very much! Now, I have to ask this: Do you happen to have this article in such form You could convert it to a PDF or something easily dowloadable? If not, then no worries, I can still download it to Word and convert it myself. However it’s just tedious and bothersome so I thought I ask first. This is a great reference and source for a up and coming exercise scientist such as myself.
http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0002024/
I’ve basically had the exact same thing on my eyes using a glorified contact lens. It’s not fun.
http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0003867/
Very interesting, thank you.
As an aside, I’ve never seen EMG data on goblet squats. How do thery compare to the back squat at various loads?
exellent post
Great, great article!
Could serve as a introductory lecture on that topic at every university.
While apparently not that popular with your average client (“I need that booty asap!” 🙂 ), these are the articles that stand out and persevere.
More of that!
In the limitations you did not mention loads selection. In exemple, If you use 80kg in the wide grip bench press and compare triceps activation during a 80kg close grip bench press, you are going to find that the close grip elicit a higher triceps activation but is it really due to the exercise itself or to a greater effort produced in the close grip bench press due to the fact that you used a heavier load, a higher percentage of the 1RM in the close grip bench press ? The problem might be even more prononced if you compare bench press and standing press. The subjects are (likely) more accustomed to bench press and thus they will be able to produce more force during this exercise compared to the standing press which requests much more pratice before you can produce full force. So the results of EMG tests might indicate a higher triceps activation during bench press compared to standing press or only a slighty higher front delts activation during the standing press but does it really relflect the truth ?
In my opinion, loads selection and the degree of practice on the exercises chosen are the biggest limitation to EMG tests. If all the tests are done on machines then the limitation due to (lack of)stability might be eliminated.
Vincent, this isn’t a limitation of EMG, it’s a limitation of study methodology. Most studies using EMG for studying glutes test either bodyweight (physical therapy studies), or identical relative loading (strength training studies). So they’d use for example a 5RM for back squats, front squats, and overhead squats, which would require different loading on all three variations, thus making the exercises equal in terms of effort requirements. In other words, a good study with sound methodology would not commit this error in study design.
Bret, does absolute EMG activity increases as muscle size increases?
For example, does someone who had 150 kg 1-rm squat and now has 300 kg 1-rm squat also increase their absolute EMG with the same relative intensity (130 kg vs. 260 kg)?
Hi Art,
There are a number of factors that influence absolute EMG activity and muscle size, specifically circumference at the muscle belly, will result in increased EMG activity. Assuming that all other things are equal, you should see the same relative intensity between those 2 scenarios you described.
Hi Bret,
certainly good written article. i want to cite your findings. how may i? is it published somewhere! definitely citation form blogs is not acceptable in most of the cases.
also about Arthur Jones (particularly the one you are talking about), does his findings ever got published?
Thanks