This article is going to seem a little “out there” for most of my readers, but try your best to understand the information as it’s very important for exercise science and I will be referring to this article as time goes on. For the past six months I’ve really delved into the literature involving structural proteins. This has been a very intriguing process. First I will discuss some important concepts in musculotendinous modeling, then I’ll delve into some exciting new research involving titin and myotilin – two exciting proteins that play a huge role in athletic performance.
Contributors to Force – The CC, PEC, and SEC
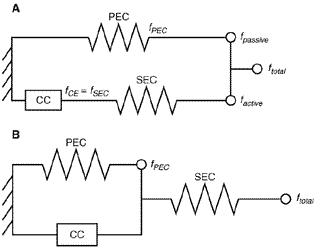
Below is a figure that will help you understand the primary components that contribute to the development of force. You will notice that this model contains three components.
The first part of the model is the contractile component which is symbolised by the square box in models A and B. The contractile component represents a conversion of electrical stimulation into force production via the actin and myosin filaments.
The second component of the model is the parallel elastic component which is symbolised by a spring- denoting its elastic like qualities. When muscle is stretched beyond resting length, the parallel elastic component contributes to tension. The structures associated with the parallel elastic component are the fascia surrounding the muscle such as the perimysium, endomysium, and epimysium, as well as the sarcolemma.
The third part of the model is the series elastic component (SEC), which once again is symbolised by a spring and occurs in series with a contractile component. When muscles contract, the series elastic component lengthens and stores energy, and therefore can contribute to total tension. The SEC is primarily the tendon which is responsible for transferring the forces developed by the contractile component or the cross-bridges to the bone and thereafter subsequent movement. These three components contribute to active and passive tension within the muscle. The active tension, via the contractile component, is mediated by the nervous system; whereas the passive tension, which comprises of the parallel elastic component and the series elastic component, are not.
What I love about this model is that it allows you to understand what’s going on at a smaller and larger scale when it comes to force production. The top picture could represent force contribution at a microscopic level, where the PEC could represent deep fascia cells, the CC could represent a single sarcomere, and the SEC could represent the titin strands. The bottom picture could represent force contribution at a macroscopic level, where the PEC could represent elastic tissues such as the deep fascia and sarcolemma, the CC could represent an entire muscle belly, and the SEC could represent the tendons. This model does a good job of illustrating the fact that active tension coincides with passive tension both parallel and in series on a smaller scale as well as a larger scale.
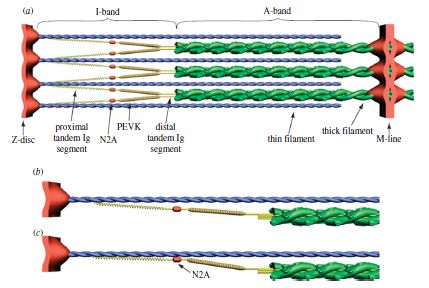
We’ve been taught that muscles produce force through cross-bridge formation (the sliding filament theory) and that muscle force gets weaker as the muscle is stretched. Many have heard of the length-tension relationship, which dictates that force will decrease as a muscle is stretched to long lengths due to the fact that less cross bridges will form. While this is true and active force does in fact diminish, passive force rises to “pick up the slack.” Below is a model of a rabbit psoas muscle.
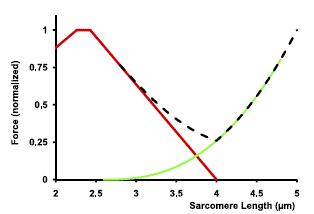
Depiction of the force-length relationship with active (red), passive (green), and total (black dashed) contribution for rabbit psoas muscle force
As you can see, the contractile force does indeed diminish, but elastic force rises as the sarcomere is stretched. Researchers have always wondered where this extra passive force is coming from, and have long suspected that it comes from a molecular protein called titin.
King Titin
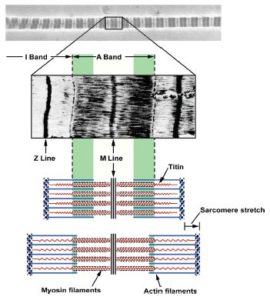
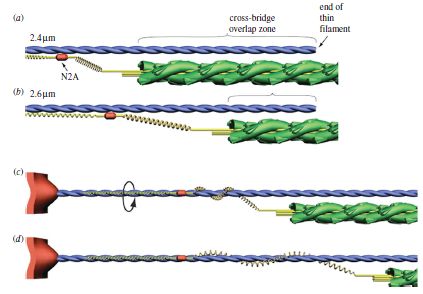
Titin is actually the largest protein in existence and consists of 34,350 amino acids! Here’s the way we used to think of titin during active stretching (think eccentric contraction):
Some brilliant researchers have been making some very good headway as to where all this passive force is generated. Last year, a researcher named Tim Leonard came up with a very good theory in the AJP – Cell Physiology, suggesting that titin actually binded to actin when actively stretched (think eccentric contraction). Here’s his model:
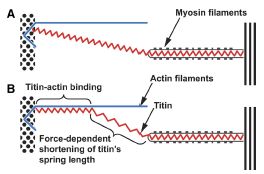
Just recently Kiisa Nishikawa published a fantastic article in The Proceedings of Royal Society B. What she proposes is that titin first binds to actin like this:
Then, titin winds up during the active contraction like this:
This model effectively explains why concentric contractions require so much more energy than eccentric contractions and why we’re much stronger eccentrically. The contractile components do all the work winding up and stretching titin when the sarcomere shortens, and then the incredibly strong and stiff titin recoils and unleashes elastic energy upon eccentric lengthening. Titin therefore provides much stability at long muscle lengths and helps protect the sarcomeres from damage. Furthermore, titin acts as a spring, so it’s not just the tendons that provide “springiness” or “elasticity”; it’s also the muscle fascia and especially the titin within the sarcomeres.
What’s very interesting is that our titin isoforms grow shorter and stiffer as we age from babies to adults (to allow us for more stability and greater force production), and powerlifters, Olympic weightlifters, and sprinters have all been shown to possess titin alterations that lead to enhanced force production. The bottom line is that training at long muscle lengths will lead to favorable adaptations which will dramatically enhance force production through stronger titin and greater elastic recoil.
Added Muscle Length
Two different phenomena have been shown to add length to muscles; prolonged lengthening and eccentric contractions. Individuals who receive casts with their muscles in long positions will see an increase in sarcomeres in series which quickly returns to normal after the cast is removed. This illlustrates how long duration stretching can add length to the muscles, but it’s a slow process. Walking downhill (pure eccentric contractions) versus walking uphill (pure concentric contractions) has been shown to result in increased sarcomeres in series in the downhill walkers compared to uphill walkers. Researchers have known this for quite some time – that eccentric contractions produced an increase in sarcomeres, but they weren’t quite sure as to why this occurred.
Furthermore, it’s been shown that training at long lengths and performing eccentrics shifts the region where a muscle develops optimal torque to longer lengths. For example, training the hamstring muscles at long lengths and with eccentrics will cause the muscles to be stronger at long lengths and weaker at shorter lengths. What explains these phenomena?
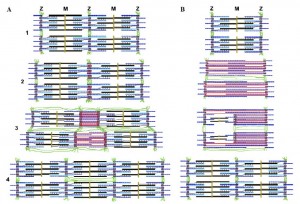
Myotilin
Lena Carlsson in Neuromuscular Disorders showed that during eccentric contractions, a protein called myotilin plays a key role in myofibrillar remodeling and the creation of new sarcomeres in series. Sarcomerogenesis can occur in two ways; through a split of a single Z-disc, or by alteration of a single sarcomere, which is shown below.
The take home point is that this new length is only added through eccentrics and this helps prevent muscles from injury. This has implications ranging from postural to athletic enhancement.
Conclusion
I realize that this material is quite complicated, but just know that full range movements that include the eccentric component are critical in the enhancement of mobility and stability. Many practitioners are aware of the motor control and neural aspects of mobility and stability, but are unaware of the structural aspects involved. I believe that this research is of enormous importance to the physical therapy and sport science fields.





Complicated but essential for a full understanding of exercise biomechanics. it proves, once again, how important full range and dynamic mobility work is.
Bret,
I have one question. There is a reason for my question and I’m not attempting to start trouble here :). My question is “how do you know that everything that you have stated is true and factual?
Rob
Short answer – I don’t. In fact, I’m positive these theories will evolve over time as more is learned.
Long answer – based on 1) email exchanges with the researchers, 2) studying the progression of the literature, 3) reading Granzier’s commentary on Leonard and Herzog’s article (and he’s the Godfather of all of this), and 4) using common sense, I believe they’re on the right path.
I can send you some of the articles if you’d like…
Bret,
If you are amicable, I’d love to get my hands on some of that research material, as I feel this line of study has some profound implications. I’d very much appreciate it.
Thanks,
Mario
Hey Bret, can you send me some of the articles you´ve read on this topic, because it´s pretty hard for me to get access to all those really interesting and important resources.
Thanks a lot!!!!!
And I thank you even more for your great work!!!!
Mathias
Guys, I’m super-swamped right now, staying at a hotel with only 30 min of internet access so maybe email me during next week at bretcontreras@hotmail.com and I’ll send you some articles. Cheers. BC
No problem, Good luck with your presentation!
M.
Yes, thank you. My point is this. On many websites, including this it has been discussed how research is often BS. Now I’m not stating that ALL research is good, however, we need a proven foundation to base our efforts. If we do not have this knowledge, they why do we prescribe the specific programs that we provide to our clients/athletes. “Common Sense” is based on proven fact or else why would it be “common sense”? If we want to incorporate eccentrics, concentrics, plyometrics, exercises at different muscle lengths, etc… the actual exercises employed may differ based on individual preference but the reason for employing the specific principles is based on proven evidence. If there is no evidence to support an application of an exercise (a) how do we know it will provide the results to a desired outcome and (b) what would be the reason to even prescribe such an exercise or principle? Research isn’t B.S. it’s the foundation for reason in our selection of training.
Rob, I totally agree. For the record I don’t think that research is BS…I think that some is of higher quality than others, incorrect conclusions can be drawn, etc., but it most certainly forms the foundation for every reason in our selection of training. We may differ in our interpretation of research and sometimes we may “reach” to attempt to be ahead of the research, but in the end we just have to evolve as things we thought we knew weren’t correct, etc. I do my best to be evidence-based, while obtaining evidence from literature, discussions with coaches, anecdotes, science, etc. And I’m certainly going to conduct good studies to try to confirm or reject various hypothesis of mine so I can add to the literature and help other coaches.
Bret,
As I have sent you research articles relating to titan-myosin cross bridging in the past I truly appreciate the article. Thank you.
Thanks Brad, glad you like it and happy that there are folks out there who find this interesting. Cheers!
So I’m assuming the benefits of EQI’s would fall under this theory, as well as DC’s extreme stretching protocols?
TJ, I would say yes. And I agree with Rob; there is no evidence for this but it’s quite easily testable. My friend Jeremy Frisch loves EQI’s and I could make a strong case for why they could be the most effective form of flexibility training. Research in this area needs to be conducted so we can learn more about it. There could be some reasons for not wanting to perform EQI’s, however, and especially DC’s extreme stretching. Overall I’m a big fan of EQI’s but I haven’t used them enough to develop protocols.
I am referring to the evidence to why we stretch, how a muscle adapt’s to become stronger, more powerful, the basis of an SCC. You are describing techniques, which may or may not have been proven to accomplish this task.
Sorry Rob, my question was to Bret.
TJ,
No apology necessary. I was then one who made the mistake.
Rob
Awesome! Long live E.Q.I. 😉
No problem. Excellent comments though. It’s so nice to see both sides of the equation
Ha. Jeremy. It was actually your articles and facebook posts that led me to that question.
Nice, well let me know if you have any questions about them!
Great stuff Bret.
Have you got any research to support the fact that eccentric contractions cause an increase in sarcomeres (not trying to catch you out I’d just like to read a few papers)?
I know Ken Nosaka out of ECU believes in it, I read a presentation he did on it but couldn’t make much sense of it!
Are these contractions accentuated eccentrics or are they performed slower than usual or just normal eccentric contractions?
Thanks,
Fionn.
Fionn, yes; I thought I mentioned them in this post. I have a couple of papers on the topic. I have so many articles I’m working on maybe it’s in a future post…yeah that’s right. I will discuss them in a future post – there are a couple. Nosaka was my friend’s supervisor – he’s very intelligent. And good question about whether or not they were accentuated – they were not; just normal eccentric contractions. Makes me wonder if accentuated would be even more effective, or less. Cheers!
So that’s why despite a pretty big stretch you can still move a lot of weight on dips… titin. Superb article, thank you for writing it.
This man backs up the article (just without the strength training).
http://www.youtube.com/watch?v=8GszNJ1NCWo
I actually think my former client Kenny Dobbs is a better dunker (not taking any credit for his freakish vert – he was like this before he came to me and I only trained him for a brief period of time): http://www.youtube.com/watch?v=OzTNc4kPQ-4
Good article, Bret!
Maybe Im been slow, hows does the vid back up the article. Great vid btw, seen it before, always wondered about this guy, how he got to jump so high, did he train, just gifted, in which way.
His program is literally 80% stretching (if not more). He’s stretching 7 days a week, 5x over the course of the day. No barbell work at all/very little emphasis on strengthening. His ROM is incredible right throughout his body, particularly in his ankles.
http://sportvideos.com/product.php?productid=1624&cat=93&page=6
This is a whole other topic; I want to write an extensive article on power production as there are plenty of factors to consider. I’ve read so many articles on the topic and could do a great job with it – but I’m so darn busy. I’m fascinated by the freaks and wondering how in the hell they jump so high.
I would put it down to there tendon elasticity.
Yeah that would be good. An article on tendons Bret.
I read an article from Kelly Baggett about this. He basicly suggested that Kadour is only a genetic freak with perfect leverages for jumping. And that the stretching probably are of no considerable effect. I have yet to see any research indicating that streching have positive effects on jumping, only short term negative effect.
It would be great if you could write an article on the matter.
Great article! I remeber Mel Siff stating something that EQI style streching give a greater streching effect on SEC and that static streching gave more effect on PEC. At least i Think so, will check it out. Do you have any thoughts on this, and maybe some practical implications?
This research also seem support the idea that hamstrings strength training at short lengths (such as leg curls) are not beneficial for eccentric strength.
Thank you for all the great articles on essential topics. With regard to the question on passive tension in a muscle, is it Titin or Collagen, I found this great article that shades some light on the history of the quest to find an answer:
Gigantic variety: expression patterns of titin isoforms in striated muscles and
consequences for myofibrillar passive stiffness
Neagoe C1, Opitz CA, Makarenko I, Linke WA.
http://www.py.rub.de/mam/py/content/kardp/03_neagoe_jmrcm_finalpublished.pdf