By Eirik Garnas
www.OrganicFitness.com
“We are survival machines – robot vehicles blindly programmed to preserve the selfish molecules known as genes. This is a truth which still fills me with astonishment.” ― Richard Dawkins, The Selfish Gene
We’ve all heard it… People complaining about bad genetics being the reason they can’t lose weight, build muscle, or get rid of their chronic health problems. When neither diet nor exercise seems to bring any results, it’s easy to start blaming something that is out of our control, and since we know that genes determine our predisposition to obesity, type 2 diabetes, and other disorders with a genetic component, mum and dad quickly end up getting the blame. But, while we’ve known for a long time that the human DNA we inherit from our parents shape our physiology and health, we’re now starting to understand that the microbial genes in our body could be even more important than the 23 pairs of human chromosomes in terms of determining our susceptibility to disease. We’re also learning that the microbial ecosystems in our body impact the hormonal and inflammatory mileu, and thereby our ability to lose weight and build muscle and strength.
The human body is a microbial world
Results from massive research projects around the world, such as The Human Microbiome Project (U.S.) and MetaHIT (EU), have made it clear that more than 75% of the cells in the human body are microbial and that the genetic repertoire of these germs is at least 100-times greater than that of the human host (1,2). Just as our human cells contain genetic material that make up our human genome, the much smaller microbial cells in and on our body contain the collection of genes that make up the human microbiome.
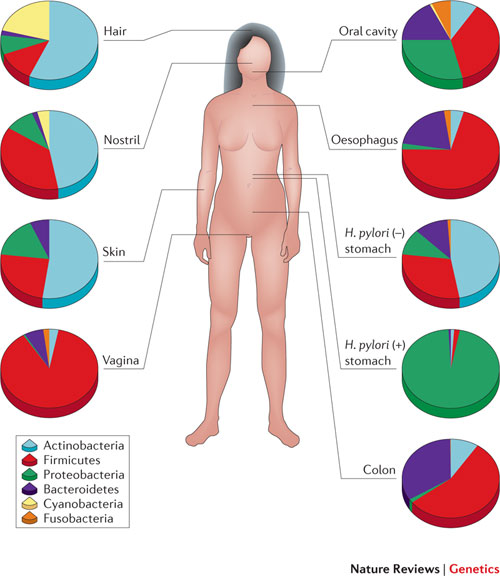
The discovery that we harbor a second genome that contributes 99% of our total genetic information is now changing the traditional perspective on health and disease. The human body is essentially an ecosystem made up of complex bacterial communities that inhabit the lungs, urogenitals, gastrointestinal tract, skin, and several other organs (3).
If these critters were just passive bystanders on our journey through life, we wouldn’t really care that much about the structure and function of the microbial rainforest that inhabit our body. However, we’re now learning that the composition of the human microbiota – all of the microorganisms that live in and on the human body – largely determines whether we get sick or not.
Microbial imbalances set us up for disease
It’s long been known that tooth decay and periodontal disease are associated with an inflammatory oral microbiota and that bacterial vaginosis and yeast infections are primarily caused by an imbalance of naturally occurring vaginal microbiota. An avalanche of studies published during the last decade have also made it clear that alterations in the bacterial communities in the lungs are closely connected to pulmonary disease (4) and that gut dysbiosis plays a role in acne vulgaris (5), type 2 diabetes (6), irritable bowel syndrome (7), and many other disorders.
Since the microbiome regulates the immune system, it’s no surprise that researchers are routinely discovering new disorders that are associated with alterations in the microbial communities in and on us. While we still have a long way to go to determine cause and effect in many disorders, it’s clear that these microbial old friends have a significant impact on our health.
Overweight and lean individuals harbor different microbes
Overweight, obesity, and weight loss are often of special interest to those that are involved in health and fitness, and in a previous article for bretcontreras.com, I discussed recent research showing how gut microbiome plays a role in weight regulation. By now it’s established that obese and lean individuals harbor different types of bacteria and that microbial imbalances can cause weight gain through several different mechanisms (8).
“The obese microbiota” seems to have an increased abundance of proinflammatory microbes, and some of these germs contain endotoxins called lipopolysaccharide (LPS) in their outer wall. People who are overweight and obese have an increased abundance of this toxin in the blood, a state referred to as endotoxemia. When LPS binds to the protein toll-like receptor 4, it triggers a set of events that leads to chronic low-grade inflammation and possibly leptin and insulin resistance and weight gain (9,10).
Beneficial bacteria boost testosterone production and athletic performance
The immunoregulatory effect of the microbiome could help explain why studies show that bacteria play a role in controlling the production of anabolic hormones that are essential for muscle growth and recovery between workouts (11,12,13). In a recent animal study, researchers found that “male mice routinely consuming purified lactic acid bacteria originally isolated from human milk had larger testicles and increased serum testosterone levels compared to their age-matched controls.” (11). And it wasn’t just a small difference, mice eating the probiotic bacterium Lactobacillus reuteri had significantly higher levels of circulating testosterone regardless of the type of diet they consumed. Given the anti-inflammatory properties of beneficial bacteria, it seems likely that these effects also extend to humans.

To be perfectly clear, nobody’s denying that human genetics play an important role in bodybuilding, strength training, and general sports performance. However, we’re now learning that the microbiome also plays a role by regulating the hormonal and inflammatory environment in our body.
We inherit the second genome from our family
The womb is not sterile
While it was previously thought that the uterus was a sterile environment, recent reports suggest that the microbiome could be seeded before birth (14,15). However, it’s not yet known which types of bacteria that are delivered to the fetus and whether they are able to colonize the unborn child.
The microbiome is seeded during birth
What we do know is that birth method has a significant impact on the microbiome. While a child who’s born through ceasaran delivery will primarily pick up bacteria from the skin of the mother, children who are born vaginally acquire bacterial communities that resemble their mother’s vaginal microbiota (16). Since the birth canal is inhabited by beneficial bacteria like lactobacillus, a natural birth helps seed the microbiota of the neonate with the right mix of microorganisms.
Children who are born through caesarean section have higher risk of asthma, allergies, ezcema, and type 1 diabetes, and one recent study also showed that caesarean delivery is associated with increased body mass in childhood and adolescence (17,18). So, giving birth the natural way is clearly the preferred option when possible, but even then there’s a chance you’re not giving the infant the best possible start on life. If the mother has some type of vaginal dysbiosis, this will clearly affect the types of bacteria that are picked up by the child during birth.
In a very interesting study in mice, researchers found that stress during pregnancy can significantly change the balance of microbes that live in the vagina and that this altered microbiota is then transferred to the infant during the birth process (19). The newborn pups who picked up the imbalanced microbiota had alterations in the brain region called the hypothalamus, and since we know that gut bacteria impact our mood, behaviour, and thoughts, this shouldn’t really come as a surprise.
Whether these mechanisms also apply to humans remains to be seen, but since it’s already established that chronic stress, antibiotics, highly processed foods, etc. can significantly alter the gut microbiota, it’s not unreasonable to think that these effects can also extend to the birth canal.
Breast milk contains hundreds of species of bacteria
After the newborn has received its first influx of critters from the birth canal and/or skin of the mother, breast milk becomes the main sources of energy unless the child is formula fed. Human breast milk contains hundreds of species of bacteria and various growth factors, but not all breast milk is the same (20). While the research on the human breast milk microbiome is still in the early stages, we’ve learned that several factors can skew the composition of bacteria found in nature’s original superfood.

Milk from women with a high body mass index contain a different and less diverse bacterial community compared with milk from normal-weight mothers, and milk from elective mothers who perform a cesarean delivery seems to contain a different bacterial community than milk samples from individuals giving birth by vaginal delivery (21). It’s also clear that use of antibiotics during lactation or pregnancy decreases the levels of good bacteria in breast milk and therefore jeopardises the health of the child (22).
Some reports suggest that microbes can travel from the mother’s gut to her breast milk, indicating that the composition of the mother’s gut microbiota could impact the types of bacteria that are transferred to the child (23,24,25,26). So, while we’ve known for a long time that breastfeeding is superior to formula feeding, we’re slowly starting to learn that it’s not just a question of breast milk vs. bottled milk, but that the types of nutrients and microbes found in human breast milk depends on the health of the mother.
The microbiome is shaped throughout life
Contagious health
And it isn’t just through the birth process and breast milk that newborns pick up bacteria that colonize the skin, mouth, gut, and the rest of the body. The microbiota is also seeded with bacteria from mum, dad, and other people through kissing, cuddling, and pre-chewed food.
It’s long been known that bacteria and viruses can be transferred from one person to another, but we’re now learning that this contagious health also could play a role in diseases that were previously considered non-communicable. If we look at the human body as an ecosystem, as we should, we also have to adhere to the basic principles of ecology. In the natural world no organisms live in isolation, but rather they interact with the ecosystems in the environment. When a child licks his fingers covered in soil or kisses his mother he’s exposing himself to microbes, and some of these germs could have an impact on his own microbial aura.

Most of the critters we’re exposed to are either killed by stomach acid or have no impact on our physiology whatsoever, but some types of bacteria such as Bacillus subtilis, a common probiotic found in soil, could take up root in the gut or transfer genes to other microbes living in colonies in the gut called biofilms. Yet other types of germs could have an impact on the skin and oral microbiota. Most of these microbes we encounter are either commensals (they benefit from colonizing our body) or mutualistic (both the microorganism and the human host benefit), but some can also be pathogenic.
This dynamic nature of the human microbiome can help explain why people living in non-westernized settings typically harbor a larger diversity of microbes compared to people in the industrialized world (27). Humans have evolved for millions of years in close contact with the vast microbial ecosystems found in soil and untreated water, and it’s only recently that we have distanced ourselves from mother nature through urbanization, sanitation, and modern hygiene. These changes have helped us combat several types of infectious diseases, but they also come with some hidden costs.
For someone who’s new to the idea of the human body as an ecosystem, the concept of contagious health might seem far-fetched. The interaction between our own microbiome and the microbial ecosystems we encounter is also very hard to measure adequately in scientific studies, and we therefore have to be cautious not to oversell the idea. However, we do have some clues to go on…
Several studies have now shown that dental caries, the most common chronic childhood disease, is in fact contagious in the sense that mothers can transmit caries-producing oral bacteria to their babies (28,29). Tooth decay is basically a bacterial infection, but do these same principles apply to other disorders that were once considered non-communicable? Since a lot of chronic health disorders are associated with microbial imbalances in the oral-. skin-, gut- and/or lung- microbiota, I believe they do, but we really don’t have enough data to say for sure. Studies have shown that germ-free animals who are inoculated with microbiota from an obese person gain more weight than animals who receive microbiota from a lean person (30), but to which degree microbes can be transferred from one person to another through kissing, uncleaned cutlery, etc. remains unclear.
The health of your own microbiome determines how sensitive you are to pathogenic microbial invaders, and it’s therefore no surprise that people with dysbiosis and low immune function are more susceptible to microbial infections and disease.
The western diet and lifestyle perturb the microbiome
While the foundation of our microbiome arrives from our family, the second genome responds to changes in diet, lifestyle, and environment throughout our lives. In the same way that human activities, such as forest fires and pollution, have disrupted the ecosystems of animals and plants that live on planet earth, antibiotics, hand sanitizers, highly processed foods, indoor living, formula feeding, caesarean sections, and other factors associated with life in the industrialized world have changed the balance of critters that inhabit the human body and disconnected us from some old microbial friends that used to make up the microbiota of our stone age ancestors.
We have the ability to manipulate our genome to an extent never previously thought possible
While it’s been known for a long time that we can change the expression of our human genes through diet and lifestyle, we’re now learning that we can alter our total genetic repertoire to an extent never previously thought possible. By manipulating the microbial communities in and on our body we’re not only changing how the microbial genes that are expressed, but also what types of germs that make up our microbiota.
Two examples of this is lactose and gluten intolerance. Most people with lactose intolerance lack the allele (one of a number of alternative forms of the same gene) that produces the enzyme to degrade lactose, and lactose intolerance is therefore often considered a permanent condition. However, several types of bacteria, such as lactobacillus found in yogurt, are able to break down lactose. Even though these probiotics don’t necessarily take up root in the gastrointestinal tract, they are able to transfer lactose digesting genes to the bacteria living in gut biofilms through horizontal gene transfer. Since bacteria are able to degrade a wide range of food ingredients, it’s no surprise that studies show that lactose intolerance can be treated with probiotics and that a large variety of bacteria are capable of utilising gluten proteins and peptides as nutrients.
While it’s still early times, dietary changes, prebiotics, and probiotics (both oral and topical) are already starting to get used in the treatment of obesity, acne, and many other health disorders.
What does all of this mean?
The expanding view of the human body as a superorganism made up of both human (eukaryotic) and microbial (prokaryotic) cells changes our perspective on the global ecosystem, human health, and genetics.
It’s increasingly being recognized that we have to consider both our human genome and the microbiome when we’re making decisions related to diet and lifestyle.
Part of the reason why some health disorders are more common in some families than others is that microbial communities are passed on from mother to child and shared between people of the same household. We should therefore aim to be healthy not just for our own sake, but also for the sake of the people around us and the children we bring into life. Since the microbiome is seeded with bacteria from our family, part of the hereditary component of health disorders characterized by dysbiosis stems from transferring imbalanced microbiota from mother to child.
Humans are very similar in terms of our human genome, but the microbiome can vary significantly from one person to another. While both of these genomes were developed in the ancestral natural environment, the microbiome is more susceptible to changes in environment and lifestyle than the human genome. The typical human microbiome of the 21st century is probably very different from that of our hunter-gatherer ancestors, and it’s likely that some old friends are now gone forever.
The general message that “Genes load the gun and environment pulls the trigger” still holds true, but we’re now learning that the genetic material is far more important than what was previously thought. It’s also clear that even if we are given a poor mix of bacteria from our family, we do have the power to change this microbiome by making the right lifestyle choices.
Some strategies that promote a healthy microbial ecosystem for you and your children
- Avoid antibiotics if possible (31)
- Don’t be too hygienic (32)
- Eat a healthy diet that includes traditionally fermented foods and sources of prebiotics (33)
- Spend time outdoors, and keep good airflow in your home (34)
- Let your kids play in the dirt, and don’t be afraid to get dirty once in a while yourself (35,36)
- Women: Perform a vaginal birth if possible and breastfeed your children
Perhaps Justin Sonnenburg, a microbiologist at Stanford, was onto something when he said that the human body is best viewed as “an elaborate vessel optimized for the growth and spread of our microbial inhabitants“…
About the author
Follow my new page on Facebook
 Name: Eirik Garnas
Name: Eirik Garnas
Website: www.OrganicFitness.com
Besides studying for a degree in Public Nutrition, I’ve spent the last couple of years coaching people on their way to a healthier body and better physique. I’m educated as a personal trainer from the Norwegian School of Sport Sciences and also have additional courses in sales/coaching, kettlebells, body analysis, and functional rehabilitation. Subscribe to my website if you want to read more of my articles on fitness, nutrition, and health.





Also: get a pet !
Very good points.
Just a small correction: it should be “milieu”.
Thx Eirik/ Bret for the post… always good info on good topics !!! good job guyz ! keep it up
Thank you Eirik for the articles here. Would you have special tips for Crohn’s disease sufferers?
Hi Sven! I don’t have any special tips for people suffering from Crohn’s disease, but there’s little doubt that rebuilding the gut microbiome should be top priority.
Hi Sven – check out Dr Natasha Campbell-McBride and The GAPS Diet – http://www.gapsdiet.com/ – she treats crohns every day with spectacular results.
Best
Karl
Resistant Starch as a means to feed gut biome is getting a lot of attention right now, my personal n=1 experience with it has certainly been very positive
http://freetheanimal.com/2013/12/resistant-primer-newbies.html
Hi Eirik,
I really enjoyed the article and your last guest post of course. Based on what you have said above do you believe that auto-immune diseases are basically a poor bacteria makeup in one or several parts of the body? If so, how would you recommend determining where in the body your bacterial makeup is poor?
You also mention a few ‘lifestyle’ factors for ensuring you have a healthy microbial ecosystem. When things get messed up and need more aggressive treatment, what would you recommend then? Or what type of practitioner should you seek for further advice?
Thank you!
Hi Nick! Yes, I believe dysbiosis is the key to understanding autoimmune disease! It’s really hard to give more individualized tips than those outlined in my articles without more information about the specific case.